Understanding Nuclear Material: The Science Behind Uranium Enrichment and Proliferation Risks
The progression of a nation’s nuclear program hinges on the complex science of uranium enrichment. At its core is the process of separating specific isotopes of uranium—a dense, radioactive element—to create material suitable for different applications. While uranium is a naturally occurring element found widely in the earth, its usefulness as energy fuel or, critically, as fissile material for a nuclear weapon, is determined by its degree of refinement. The most valuable isotope for sustaining a nuclear chain reaction is U-235, which is present in minute quantities in natural uranium ore.
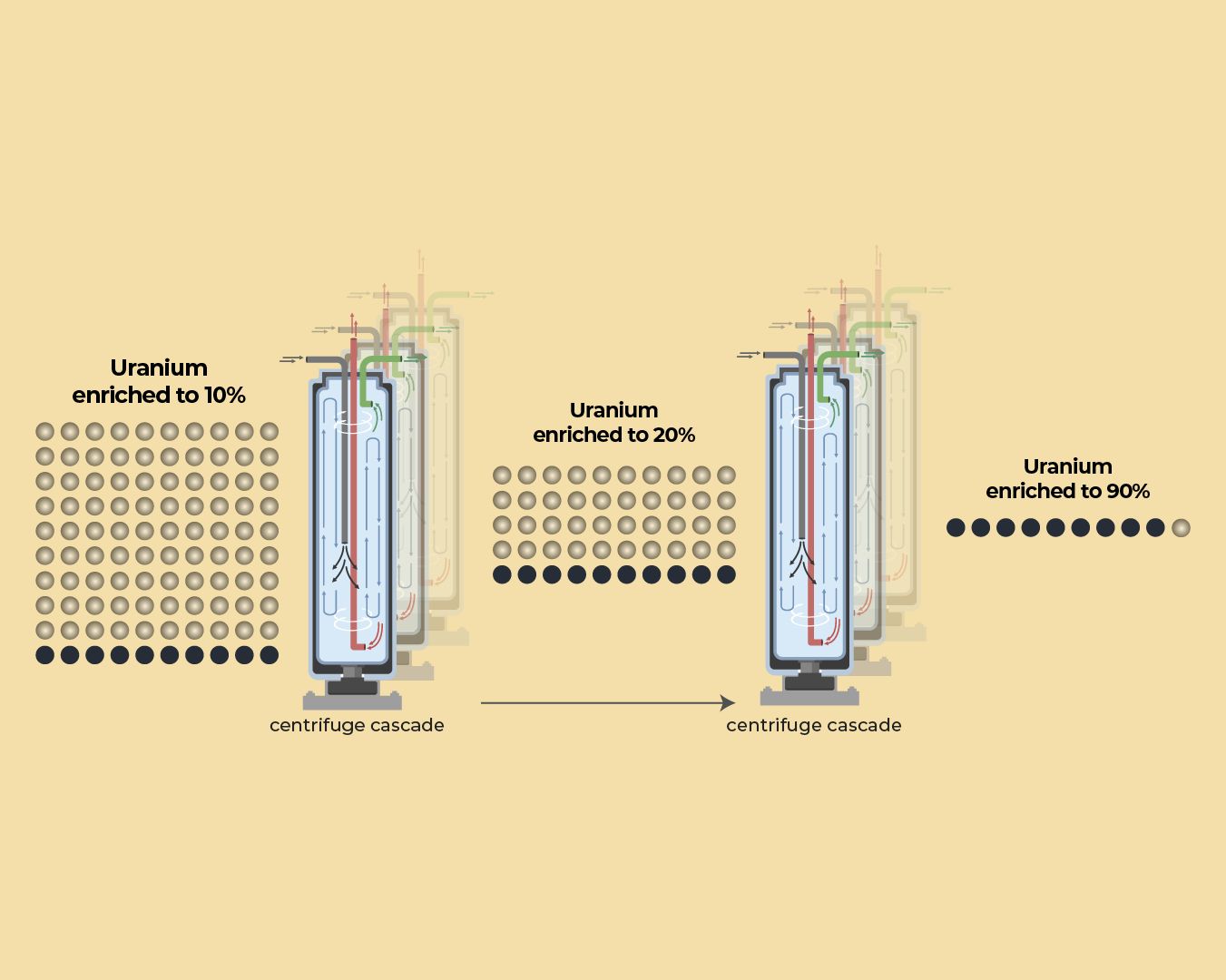
The industrial process designed to make nuclear power generation possible involves taking natural uranium and enriching it. This process selectively boosts the concentration of U-235 relative to U-238. For civilian power plants, enrichment typically targets a moderate level, usually between three and five percent U-235. However, achieving the levels required for a nuclear explosive device demands a vastly different and much higher concentration, near 90 percent U-235. This technical distinction forms the crux of global nuclear concerns regarding proliferation.
These capabilities have significant geopolitical implications, as nations possessing the expertise and material to elevate uranium enrichment could dramatically alter regional stability. When discussions occur regarding the status of advanced nuclear programs, experts frequently assess the time frames and technical hurdles involved in reaching weapon-grade material. The control and understanding of enrichment technology are thus viewed by international bodies as paramount to maintaining global non-proliferation efforts.
The Science of Isotope Separation
To provide context, understanding the isotopic makeup of uranium is key. Natural uranium consists primarily of U-238, the isotope that is the heaviest and least radioactive. U-235 makes up a tiny fraction of natural reserves, while U-234 exists in even smaller amounts. The enrichment process is essentially an advanced form of physical separation that isolates the energy-rich U-235 atoms. This ability to concentrate one specific isotope from a mixture is what separates a peaceful energy initiative from a potential arms race.
Global Implications of Enrichment Control
The global supply chain for uranium processing is concentrated in only a handful of exporting nations. As a result, the ability to process and enrich this material remains a critical focal point in international diplomacy. Discussions surrounding nuclear agreements often revolve around ensuring that enrichment activities remain solely dedicated to peaceful civilian energy purposes, thereby limiting the pathway toward acquiring weapons-grade fissile material. The technical barriers and required infrastructure for such a pivot—from low-level enrichment to weaponization—are the subjects of intense technical and diplomatic scrutiny.